Page 9 - reflections_newsletter
P. 9
REFLECTIONS
Dyslipidaemia
Dyslipidaemia Global Newsletter #11 2026
Evolocumab in patients without a previous myocardial infarction or stroke. Dyslipidaemia
Bohula EA, et al. N Engl J Med. 2026 Jan 8;394(2):117-127.
LDL-C is a well-established, modifiable risk factor for CVD. In recent years, lowering LDL-C levels with PCSK9 inhibitors, such
as evolocumab, has been proven to significantly reduce the risk of CV events. However, this treatment has historically been
studied almost exclusively in secondary prevention settings. For example, earlier pivotal studies like the FOURIER trial focused
on adults with established ASCVD, specifically those who had already experienced a prior MI, stroke, or symptomatic peripheral
artery disease. Until now, the effect of PCSK9 inhibition on preventing a first major CV event in high-risk patients who had not yet
suffered an MI or stroke remained largely unknown.
The VESALIUS-CV trial evaluated whether evolocumab could effectively lower the risk of a first major CV event in this previously
unstudied demographic. The researchers conducted a large international, double-blind, randomised, placebo-controlled trial
involving 12,257 patients. Eligible participants were high-risk individuals with atherosclerosis or diabetes who had no history of MI
or stroke, and who were already receiving stable, optimised baseline lipid-lowering therapy. Patients were randomly assigned to
receive either subcutaneous injections of evolocumab (140 mg every two weeks) or a matching placebo. The investigators then
tracked the patients over a median follow-up period of 4.6 years, looking primarily at two composite endpoints: a 3-point major
adverse cardiovascular event (MACE: death from coronary heart disease, MI, or ischaemic stroke) and an expanded 4-point
MACE (which added ischaemia-driven arterial revascularisation). The authors also conducted a lipid substudy in which they
evaluated the change in LDL-C and other atherogenic lipids from baseline as an exploratory endpoint.
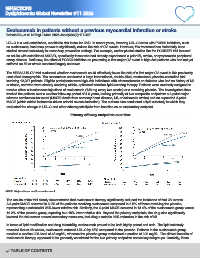
Primary efficacy endpoints over time
MACE, major adverse cardiovascular event.
The results of the trial clearly demonstrated that evolocumab therapy significantly reduced the incidence of first CV events.
A 3-point MACE occurred in 6.2% of the patients receiving evolocumab compared to 8.0% of those receiving the placebo,
representing a substantial 25% lower relative risk. Similarly, the 4-point MACE occurred in 13.4% of the evolocumab group versus
16.2% of the placebo group, equating to a 19% lower relative risk. Beyond the primary endpoints, the drug also significantly
lowered the risk across several secondary measures, including a notable 36% reduction in the risk of MI.
In terms of lipid modification and drug tolerability, evolocumab proved to be both highly potent and safe. The lipid substudy
revealed that at 48 weeks, evolocumab reduced LDL-C by 55% compared to the placebo. Patients in the evolocumab group
reached a median LDL level of 45 mg/dL, whereas the placebo group maintained a median of 109 mg/dL. The clinical benefits of
evolocumab therapy appeared to be generally consistent for the two primary endpoints across key subgroups. Crucially, these
TABLE OF CONTENTS