Page 11 - reflections_newsletter
P. 11
REFLECTIONS
Dyslipidaemia
Dyslipidaemia Global Newsletter #11 2026
TARGETS AND BIOMARKERS Dyslipidaemia
Intensive LDL cholesterol targeting in atherosclerotic cardiovascular disease.
Lee YJ, et al. N Engl J Med. 2026 Mar 28. Online ahead of print.
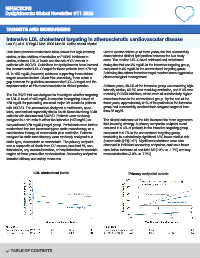
Data from previous randomised trials shows that high-intensity Over a median follow-up of three years, the trial successfully
statins, or the addition of ezetimibe or PCSK9 inhibitors to demonstrated distinct lipid profiles between the two study
statins, reduces LDL-C levels and the risk of CV events in arms. The median LDL-C level achieved and maintained
patients with ASCVD. Guidelines for dyslipidaemia have lowered during the trial was 56 mg/dL in the intensive targeting group,
the recommended LDL-C target for these patients from <70 mg/ compared to 66 mg/dL in the conventional targeting group.
dL to <55 mg/dL; however, evidence supporting these stricter Achieving the stricter intensive target required more aggressive
targets remains limited. Given this uncertainty, there exists a pharmacological management.
gap between the guideline-recommended LDL-C target and the
implementation of this recommendation in clinical practice. At three years, 48.4% of the intensive group was receiving high-
intensity statins, 66.6% was receiving ezetimibe, and 2.3% was
The Ez-PAVE trial was designed to investigate whether targeting receiving PCSK9 inhibitors, which were all substantially higher
an LDL-C level of <55 mg/dL is superior to targeting a level of rates than those in the conventional group. By the end of the
<70 mg/dL for preventing recurrent major CV events in patients three years, approximately 61% of the patients in the intensive
with ASCVD. The researchers designed a multicentre, open- group had successfully reached their assigned target of less
label, randomised superiority trial in South Korea involving 3048 than 55 mg/dL.
patients with documented ASCVD. Patients were randomly
assigned in a 1:1 ratio to either the intensive (<55 mg/dL) or The clinical outcomes of the trial favoured the more aggressive
conventional (<70 mg/dL) target group. Participants were further lipid-lowering strategy. A primary composite endpoint event
randomised into two treatment types: statin monotherapy or a occurred in 6.6% of patients in the intensive targeting group
combination therapy of rosuvastatin plus ezetimibe. Patients compared to 9.7% in the conventional targeting group,
assigned to statin monotherapy were randomly assigned to a translating to a statistically significant 33% lower relative risk
statin type, rosuvastatin or atorvastatin. The primary endpoint (hazard ratio [HR] 0.67). Significant reductions were also
was a composite of death from CV causes, non-fatal MI, non- observed in individual secondary endpoints, such as a lower
fatal stroke, any revascularisation, or hospitalisation for unstable cumulative incidence of non-fatal MI (0.8% vs. 1.7%) and any
angina at three years after randomisation. Secondary endpoints revascularisation (4.8% vs. 7.5%).
included efficacy and safety measures.
LDL cholesterol levels Primary endpoint events
TABLE OF CONTENTS